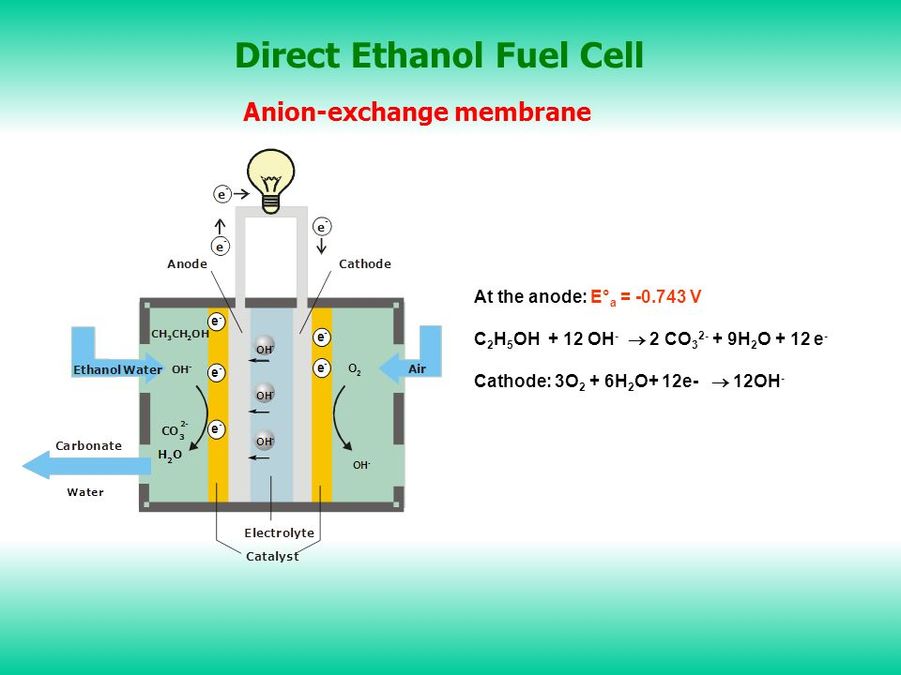
Ethanol Fuel
The diagram below shows how direct ethanol fuel cells (DEFC) use the oxidation of ethanol to catalyse the production of energy for motor-vehicles. Ethanol is pumped in to the fuel cell where it undergoes an oxidation reaction at the anode releasing electrons, water is produced as a by product which binds with water to produce carbonate water. The released electrons travel from the anode along an external circuit producing the energy for the motor. Protons (H+) which are produced in the anode reactions moves across an exchange membrane to react with oxygen at the cathode to produce hydroxide. These hydroxide molecules are then transported across to the anode where they take part in the oxidation reaction